


healthcare professional
Please refer to your local KOSELUGO® label for full prescribing information.*


*There are special safety considerations for the use of KOSELUGO®, please refer to the SmPC for full safety information.
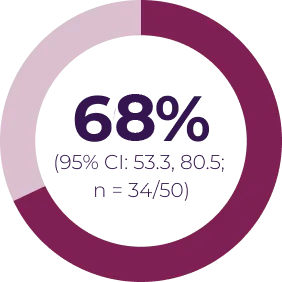
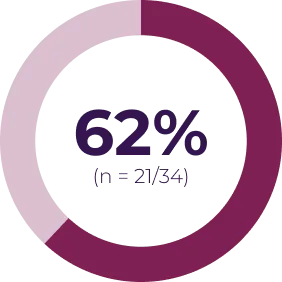
Most adverse events can be managed by dose interruption, reduction, or discontinuation. In Phase I of the SPRINT trial (N = 24), 13 patients required dose reduction for toxicity, and 3 patients discontinued treatment due to an adverse event possibly related to treatment. In SPRINT Phase II Stratum 1 (N = 50), 16 patients had ≥ 1 dose reduction and 5 patients discontinued treatment due to an adverse event possibly related to treatment.3
KOSELUGO® is a selective inhibitor of mitogen-activated protein kinase kinases 1 and 2 (MEK 1/2). KOSELUGO® blocks MEK activity and the rat sarcoma (RAS)- rapidly accelerated fibrosarcoma (RAF)-MEK- extracellular signal-regulated kinase (ERK) pathway. Therefore MEK inhibition can block the proliferation and survival of tumour cells in which the RAS-RAF-MEK-ERK pathway is activated.1,4

CI, confidence interval; ERK, extracellular signal-regulated kinase; GDP, guanosine diphosphate; GTP, guanosine triphosphate; MEK, mitogen-activated protein kinase kinase; NF1, neurofibromatosis type 1; PN, plexiform neurofibroma; RAF, rapidly accelerated fibrosarcoma; RAS, rat sarcoma viral oncogene homologue.
*Please refer to the below KOSELUGO® labels for full prescribing information:
KSA: AstraZeneca AB. KOSELUGO® (selumetinib) Summary of Product Characteristics. 2024.
GCC: Koselugo™. Prescribing Information.